Orthopedic Interlocking Nail Tips for Global Buyers on Quality
In the ever-advancing field of orthopedic surgery, the quality of instruments can significantly impact patient outcomes. Dr. Emily Harris, a renowned expert in orthopedic trauma, once stated, "A dependable Orthopedic Interlocking Nail is critical for successful fracture stabilization." This highlights the importance of selecting the right materials and designs for these vital tools.
Orthopedic Interlocking Nails are crucial for managing complex bone fractures. These devices provide stability and alignment, allowing for proper healing. However, not all products on the market meet the highest standards. Increasingly, global buyers face challenges in sourcing quality components.
As the demand for reliable orthopedic implants grows, it’s essential to stay informed. Potential buyers should carefully evaluate manufacturers' reputations and material quality. Mistakes in this area could lead to complications post-surgery. Therefore, transparency in the production process should be a priority for any supplier. A well-informed choice can ultimately make a difference in patient care and recovery time.
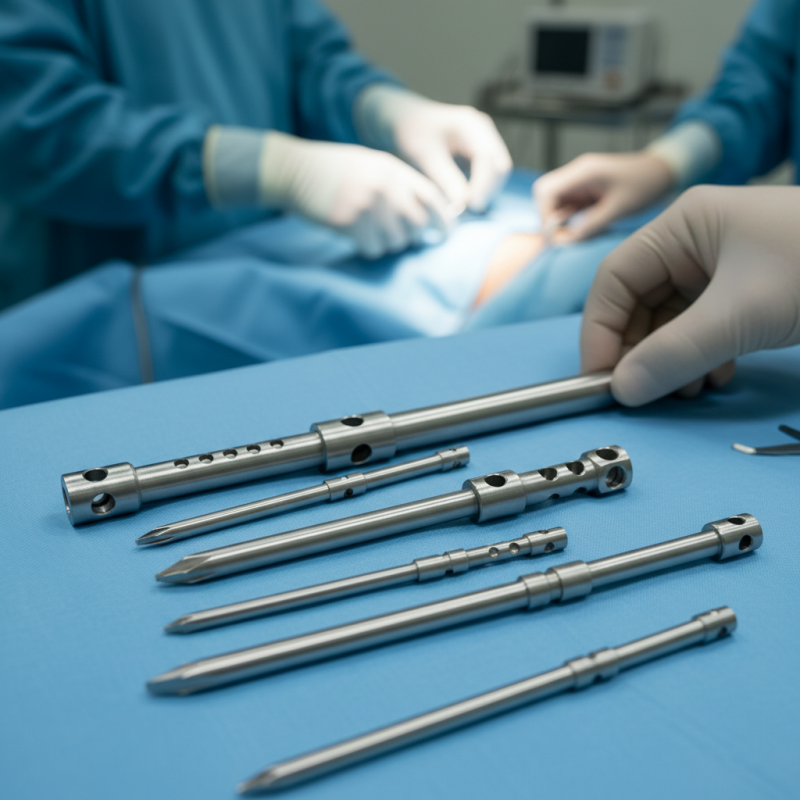
Orthopedic Interlocking Nail Design: Understanding Structural Integrity
Orthopedic interlocking nails play a crucial role in ensuring structural integrity during bone healing. Understanding their design is essential for both healthcare professionals and manufacturers. These nails are crafted from biocompatible materials, allowing for optimal integration with the natural bone. The design must support the dynamic forces experienced by the limb during recovery. This requires precise engineering and material choice to prevent complications.
While interlocking nails are effective, challenges remain in their implementation. Proper alignment and fixation are vital to prevent mechanical failure. Misalignment can lead to delayed healing or malunion, complicating the recovery process. Surgeons must carefully consider the patient's unique anatomy when selecting nail sizes and configurations. This personalized approach enhances the chances of successful outcomes.
Despite advancements in technology, there is always room for improvement in interlocking nail design. Ongoing research into new materials and methods is essential. Ensuring that these devices cater to the diverse needs of patients remains a priority. Engaging with surgeons can provide insights into real-world challenges they face. Incorporating feedback could lead to innovative solutions that enhance the overall effectiveness of these orthopedic devices.
Orthopedic Interlocking Nail Tips for Global Buyers on Quality
| Feature | Description | Material | Load Capacity (kg) | Manufacturer Country |
|---|---|---|---|---|
| Interlocking Mechanism | Provides stable fixation of fragments | Titanium Alloy | 120 kg | Germany |
| Design Type | Cannulated and Solid Nails | Stainless Steel | 150 kg | USA |
| Surgical Technique | Minimally Invasive | Carbon Fiber Reinforced | 180 kg | Japan |
| Length Options | Available in various lengths | Aluminum Alloy | 200 kg | Italy |
| Radiopacity | Visible under X-ray | Medical Grade Steel | 220 kg | Sweden |
Mechanical Properties of Orthopedic Nails: Analyzing Tensile and Yield Strength
When analyzing the mechanical properties of orthopedic nails, tensile and yield strength are crucial metrics. Tensile strength measures how much force a material can withstand while being stretched or pulled. This property directly impacts the nail's ability to maintain stability during the healing process. Yield strength, on the other hand, indicates the point at which a material begins to deform permanently. Understanding these properties helps ensure patient safety and surgical success.
Different materials exhibit varying tensile and yield strengths. For instance, titanium alloys, commonly used in orthopedic applications, offer a good balance of strength and weight. However, it’s essential to consider that not all manufacturers adhere to strict quality control measures. Variability in production processes can lead to inconsistencies in strength. This raises concerns about the reliability of certain products in delivering the expected performance.
It's important for buyers to scrutinize the specifications provided by manufacturers. Transparency in mechanical properties should be a non-negotiable standard. While some materials might perform well in laboratory settings, real-world conditions can differ significantly. Evaluating the mechanical performance of orthopedic nails across diverse scenarios remains a pivotal area for further research and development.
Global Standards for Orthopedic Implants: ISO and ASTM Compliance Explained
When discussing orthopedic implants, understanding international standards is essential. ISO and ASTM are key players in ensuring quality and safety. ISO, or the International Organization for Standardization, sets precise benchmarks. These benchmarks guide manufacturers in creating safe, effective products.
ASTM, or the American Society for Testing and Materials, provides specifications for materials and testing methods. Compliance with these standards helps ensure that implants perform under stress. It’s critical for manufacturers to engage with these compliance protocols. Without strict adherence, the risk increases for complications during surgeries.
Quality in orthopedic implants cannot be taken for granted. While many adhere to these standards, some may overlook the importance of continued testing and validation. Buyers need to scrutinize certifications carefully. The promise of compliance is not always a guarantee of quality. Thus, clear communication and understanding between buyers and manufacturers are vital for safeguarding patient health.
Quality Control Measures in Orthopedic Manufacturing: Ensuring Safety and Efficacy
In orthopedic manufacturing, quality control is paramount. The safety and efficacy of interlocking nails hinge on rigorous testing and adherence to regulatory standards. Manufacturers must implement various quality control measures throughout the production process. Raw materials undergo detailed inspections before acceptance. Routine audits ensure compliance with established protocols.
The assembly line also plays a critical role. Each stage, from machined components to final packaging, requires close monitoring. Trained professionals assess dimensional accuracy and surface finish. However, variability can arise due to machine calibration or human error. Continuous training and updates can address these challenges.
Product testing is essential. Interlocking nails must withstand multiple stress tests. Even small defects can lead to catastrophic failures. Therefore, some manufacturers may overlook minor flaws during inspections, which can be a ticking time bomb. A culture of transparency and accountability could enhance outcomes. Seeking feedback from medical professionals can guide improvements. Quality control isn't just a checkbox; it's a commitment to patient safety.
Market Trends for Interlocking Nails: Demand Insights and Future Projections
The orthopedic interlocking nail market is evolving rapidly. Demand is rising globally as more patients require orthopedic surgeries. Interlocking nails are crucial for stabilizing fractured bones. Surgeons appreciate their effectiveness and reliability in providing long-term solutions.
Recent trends indicate a shift towards more advanced materials. Innovations focus on lighter yet stronger options. Surgeons are seeking nails that reduce surgery time and enhance recovery rates. There is a growing emphasis on biocompatibility, ensuring that materials do not cause adverse reactions. Despite this progress, quality control remains an area for improvement. Variability in production standards can lead to inconsistent outcomes.
Additionally, the global market faces challenges. Regulatory standards differ significantly across regions. This inconsistency can hinder access to high-quality products. Buyers must remain vigilant and informed, ensuring they choose reliable suppliers. Staying updated with market trends is essential for making informed decisions. Adapting to these changes can ultimately enhance patient care and surgical success rates.





